Anyone?
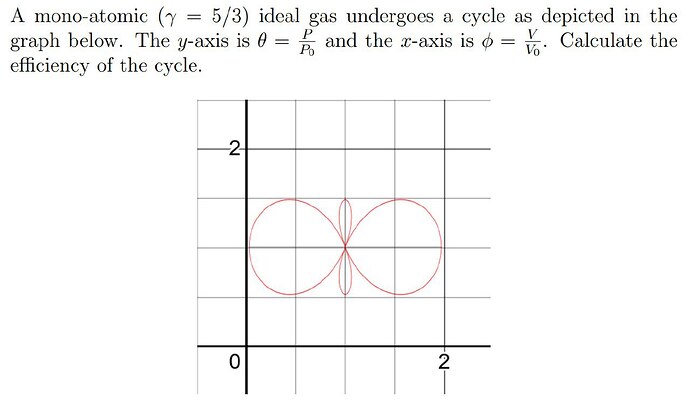
what’s the direction of a cycle?
direction is clockwise…
The solution starts very similarly to what Ersultan provided in Entropy-Temperature graph problem, try to think of it this way.
I am not able to figure it out… ![]()
what answer are you getting? the answer given is: 0.1493
The work of a gas is obtained via integrating the 1st thermodynamics law
The efficiency is a fraction between A and positive heat Q_+, so we should calculate the latter:
So we have to find points where \delta Q changes its sign, and these are simply the points where infinitesimally small adiabatic processes are proceeded, or, in other words, the points where the slope is
I’m not so sure how to find numerical values of these points. This cycle looks like mutually perpendicular lemniscates of Bernoully, so maybe we can somehow go into polar coordinates?
Pretty cringe problem tbh
This was from modified from Rudolf Ortvay 2015 problem
Yeah polar coordinates is the way to go i believe, considering we can make an equation of curve (on wolfram)